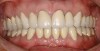
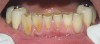
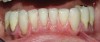
You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Tooth surface loss1 and tooth wear2 are interchangeable terms for a process that occurs both physiologically3 and pathologically2 and results in non-carious lesions. The results can produce unacceptable esthetics, compromise oral functions, cause pain and sensitivity, and negatively impact quality of life. Tooth surface loss is studied less in the United States as compared to Europe and Australia, and, currently, there is neither an evidence-based clinical protocol on whether or not to treat the resulting non-carious lesions nor an established protocol on how to treat them.4,5
Dental structure is a mineralized tissue that undergoes a dynamic process of remineralization and demineralization in the oral cavity. In the presence of supersaturated and normal functioning saliva, calcium and phosphate ions leach from the dental structures. These are then taken up in the presence of fluoride, thus maintaining a homeostasis. In response to chemical challenges and mechanical adversities, such as erosion, abrasion, attrition, and abfraction, the irreversible loss of mineral components from dental tissue occurs (Figure 1).
Tooth surface loss due to microbiological activity results in carious lesions. Tooth surface loss through mineral dissolution is known as erosion, as in the case of chemical agents or chelators acting on plaque-free tooth surfaces6 (excluding bacterial plaque acid). Erosion does not have any direct association with mechanical or traumatic actions or carious lesions produced by bacterial acid. However, erosion often coexists with attrition, abrasion, and abfraction. Attrition is the tooth tissue loss caused by antagonistic tooth-to-tooth physical contact; abrasion is the physical wear as a result of mechanical processes involving foreign abrasive substances;7 and abfraction is the physical wear as a result of tensile or sheer stress usually at the cemento-enamel region.8 The involvement of different kinds of non-carious dental lesions may make it difficult to determine the cause of tooth surface loss.
Classification of Non-Carious Lesions Due to Tooth Surface Loss
The following four categories are the general classifications of tooth surface loss. However, in most cases, a combination of these factors results in tooth surface loss and poses diagnostic and etiological challenges.9
Erosion
Tooth surface loss by erosion occurs due to acidity and the erosive potential10 in the oral cavity from intrinsic or extrinsic acid that has exceeded the buffering capability and neutralizing functions of normal saliva and salivary proteins. The term "corrosion" has also been used to describe tooth surface loss caused by chemical and electrochemical actions.8 Dental erosion occurs when the rate of demineralization exceeds the rate of remineralization of the calcified dental tissue. The hydroxyapatite crystals in enamel dissolve at the critical pH level of 5.2 and in dentin at a level of 6.9. In a normal situation, tooth structure is remineralized with the formation of hydroxyfluoroapatite crystals, which are more resistant to acidic attacks, but frequent acid exposures increase the demineralization of tooth structures.
Saliva is the most important biological factor that prevents erosion. Neurological stimuli due to acid increases stimulate salivary production thereby increasing the buffering capacity (bicarbonates), diluting and clearing acid from the dental surfaces, and maintaining homeostasis of the oral cavity. A supersaturated concentration of calcium and phosphate in saliva promotes remineralization of the demineralized tooth surfaces.
Saliva also plays a role in acquired pellicle formation, which acts as a barrier that prevents acid from contacting tooth surfaces. Other factors preventing erosion include tooth structure and position of the tongue.11
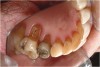
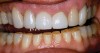
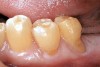
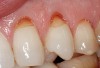
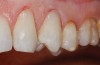
Characteristics of damage to tooth structures caused by acidity include wear on the occlusal surfaces of molars and a saucer shape on the cusps of molars (perimolysis).12 The erosion can vary from minor and subtle changes on the tooth surface, ie, loss of luster, a dull or matted look, to cupping on occlusal surfaces or incisal edges, which may extend to dentin. Usually, the erosive lesions demonstrate greater width than depth. When combined with abrasive forces or stresses, the tooth surface loss may occur at a faster rate (Figure 2, Figure 3, Figure 4 and Figure 5).
Abrasion
Abrasion is tooth surface loss due to friction caused by the abrasiveness of toothpaste and toothbrushes, which is necessary to remove biofilm and stains and to polish tooth surfaces. The rate of loss depends upon the degree of abrasivity, magnitude of force, contact time, and frequency of physical contact (Figure 6). Abrasives in toothpastes include silicates, alumina, calcium phosphate, and calcium carbonate. Manly showed non-significant changes caused by the toothbrush13 but that the hardness of the brush and the concentration of abrasive are important influences on wear caused by dentifrice abrasives and that the degree of influence depends on the abrasive.14-16 Toothpaste slurry abrasivity affects abrasion and tooth surface loss,17 and a larger amount of slurry can be produced with softer rather than stiffer toothbrushes. However, some research has not found a correlation between toothpaste and cervical lesions.18 Abrasive lesions have greater axial depth than the gingivo-occlusal distance and may possess sharp angles.
Attrition
Attrition is tooth surface loss due to mechanical forces of tooth-to-tooth contact19 or tooth-to-restoration contact. In the absence of normal occlusion, attrition may be prominent on natural teeth which oppose restorations that are harder than typical enamel or dentin. If the teeth move vertically upon occlusal loading, the wear may be seen at the proximal surfaces of the adjacent teeth. Thus, attrition occurs in the occlusal and proximal surfaces and incisal edges.
The mechanical forces affecting attrition include chewing, deglutition, and parafunctional habits. If these forces go unchecked, the enamel will develop wear facets that are usually flat and well circumscribed in both opposing arches (Figure 7).
Abfraction
Abfraction is the result of the static and cyclic loading of biomechanical forces on the teeth. The consequences of biomechanical forces, which affect the weakest part of the tooth, ie, the cemento-enamel junction (CEJ), depend on the magnitude, duration, direction, frequency, and location of the forces. As a result, the enamel or dentin may chip away, creating non-carious lesions in the cervical area20-22 that are usually V-shaped notches (Figure 8). Most of the data on abfraction is based upon engineering studies of the tooth or just the crown and a portion of the root. However, investigations involving periodontal ligaments and alveolar bone have cast reservations on the fundamental basis of abfraction.23
Common Causes of Non-Carious Lesions Due to Tooth Surface Loss
Bruxism/Clenching
Bruxism is the habitual, non-functional grinding movement of the masticatory system (mandibular parafunction) in eccentric position, as well as canine and incisal guidance.24 Bruxism causes a significant rate (three to four times the normal wear of 10 µm to 20 µm per year24) of attrition, resulting in structural changes of the tooth morphology with the formation of deep grooves, striations, and wear facets.25 Clenching is considered "silent bruxism" in centric occlusion without lateral or forward movements. The tooth wear is more severe in the anterior teeth as compared to the posterior teeth.24
Pathology
Diseases that lead to tooth structure loss include those in which the natural formation of enamel and dentin is compromised (eg, enamelogenesis imperfecta and dentinogenesis imperfecta). When the mineral density, level of mineralization, and hardness are low and the teeth are not capable of withstanding the normal physical and chemical challenges in the oral cavity, tooth surface loss occurs. The rate of tooth surface loss may increase when the protective factors of saliva, which maintains the homeostasis in the oral cavity, are diminished due to salivary hypofunction and dysfunctions, as in Sjögren's syndrome, rheumatoid arthritis, and radiation to the head and neck region.
In salivary hypofunction, the lack or decrease of saliva increases the friction and effectiveness of the abrasivity of toothpaste and toothbrushing. The site-specific tooth surface loss reflects the normal protective role of serous saliva and salivary pellicle in a site-specific manner.26 Also, involuntary regurgitation and chronic regurgitation such as in hiatal hernia and gastroesophageal reflux disease (GERD) negatively affect the oral cavity because the refluxate (intrinsic acid) is low in pH (pH < 2) and readily dissolves the tooth minerals. Dental erosion can be considered an atypical manifestation of GERD. Voluntary regurgitation in cases of anorexia nervosa and bulimia also introduces gastric refluxate into the oral cavity and causes degradation of the dental structures.27-31 Additionally, various medications with anti-cholinergic side effects (eg, psychiatric, antihypertensive, a combination of medications, etc) cause salivary hypofunction. The chronic use of medications that are low in pH (liquid, chewable tablets, or inhaler forms) has a detrimental effect on teeth and can potentially cause erosion.
Dietary and Lifestyle Habits
Frequent retention of extrinsic acids, especially citric and acetic acids in the form of drinks and food, favors the dissolution of tooth minerals and decreases microhardness. Because pH is not exclusively dependent, the "erosive potential" of food and drinks should be assessed by mineral content, triturable acidity (buffering capacity), and calcium chelation properties in reducing the microhardness of tooth structure.32,33 Food and drinks containing citric acid may be more erosive than those containing phosphoric acid.34
The current trend toward healthier food and drinks that are acidic may lead to increased tooth surface loss. Diet soft drinks that are low in calories and sugar can be poor alternatives when it comes to tooth surface loss because of their acidity and erosive potential. Also, drinking acidic (pH ≈ 3) sports beverages, especially after engaging in sports activities when the oral cavity is dehydrated, can increase the rate of tooth surface loss.
Cultural and ethnic foods that are hard to chew, dried, or fibrous, as well as certain preparation methods, can also adversely affect the amount of tooth wear.
Repeated behavioral patterns, eg, smoking a pipe, chewing pens, or using teeth to open beverage bottles, cause tooth wear. Stressful working conditions, working in factories that use acids (eg, a battery factory), working in a noisy environment, swimming in gas-chlorinated pools with low acidity and no buffering capability, occupational wine tasting, etc, can all cause tooth surface loss. Playing certain musical instruments also can contribute to specific patterns of tooth wear.
Toothbrushing and Toothpastes
Brushing the teeth in the demineralized state, after exposure to acidity, has the cumulative effect of initiating tooth wear and ultimately removing tooth structure.35 Toothbrushing in a horizontal direction causes more cervical wear than vertical brushing.36 In addition, the type of toothbrush bristles and toothpaste used exerts mechanical forces that can abrade tooth surfaces. Sharper, pointed toothbrush filaments, a higher number of tufts, dense packing, and splay on toothbrush loading37 are factors that are more likely to abrade hard and soft tissues and restorative material, as opposed to rounded tips38 and a low number of low-density tufts.
Abrasives in the toothpaste are necessary to remove biofilm and stain from the reachable tooth surfaces and to polish them. However, radioactive dentin abrasivity (RDA)39 and radioactive enamel abrasivity (REA) of the toothpaste may influence the level of enamel or dentinal wear with repeated use of the toothpaste.40 It is interesting to note that the RDA is not required by law to be on the label of currently marketed products.
Craniofacial Complex
A deviation in the vertical direction, a small angle between the mandibular–palatal planes, and a small gonial angle also play a role in tooth wear.41 The bite force and pressure applied by the mandible can be tremendous because the masseter muscle is one of the strongest in the human body, depending on the craniofacial complex. The more square the face, the more surface area for the attachment of the muscle and the more exertion of bite force and pressure, resulting in increased tooth wear.
Iatrogenic Dentistry
When the hardness of the restorative material is greater than the natural opposing teeth, wear of the natural teeth will occur in absence of bite corrections and occlusal adjustments. These problems can usually be seen locally in the teeth opposing the restoration but can be generalized as well.
Aging
Tooth surface loss and tooth wear occur naturally with age, beginning even before the tooth reaches the occlusal plane.3,42 During the functional life of a tooth, cusps flatten, wear facets develop, enamel volume is lost from the tooth surfaces, and dentin is exposed. With the increased longevity of the human population, increased retention of teeth along with gingival recession gives rise to more tooth surface exposure, thus greater potential for formation of non-carious lesions. The elderly population whose teeth are retained for longer periods of time and who have increased exposure of root surfaces may be at higher risk of developing erosive lesions.43
Effects of Tooth Surface Loss
The effects of tooth surface loss vary depending on the severity of the tooth tissue loss. These effects include loss of thickness of enamel and exposure of the underlying dentin, translucency, and yellowing. Also, the occlusal surfaces may display wear facets, or the cusps may flatten.
When tooth surface loss and tooth wear continue, the exposure of dentinal structures leads to tooth sensitivity. However, in some cases, such as the formation of a smear layer, secondary dentin, dental sclerosis, or even burnishing actions of toothbrushes, there may not be any hypersensitivity.44-46
Loss of tooth height may lead to a decrease in the vertical height, the consequences of which can be felt on the temporomandibular joint and can also lead to difficulty in eating and chewing. Rapid loss of tooth surfaces may be a potential threat to the pulp and the integrity of the teeth, and loss of tooth structure in anterior areas can result in unfavorable esthetics. Moreover, the cost to maintain and restore dentition may add an extra burden to the patient. Thus, tooth surface loss potentially has various effects on function, esthetics, and overall quality of life.
Management
Management of the complex etiological factors of tooth surface loss is of the utmost importance to prevent further loss. Restorative dentistry provided without such management is frequently unsuccessful47 and no evidence-based guidelines or studies exist for the prevention or treatment of tooth surface loss.4 It is incumbent on dentists to become cognizant of the reasons for tooth surface loss, because this will help them inform patients of the benefits to be gained by restoring such deficient areas.48
Preventive Methods
Charting the teeth using a tooth surface loss index (Table 1 and Table 2), digital photography, digital surface mapping, and casts aid in monitoring and measuring the progression of tooth wear and assessing the effectiveness of the preventive program.49
Medical conditions (eg, gastric regurgitation, salivary gland diseases, chronic renal failure) that are contributing to a patient's tooth surface loss should be addressed and managed accordingly. When saliva cannot buffer the intrinsic or extrinsic acidity in the oral cavity in salivary hypofunction conditions, use of the sialagogues (pilocarpine HCl or cevimeline HCl) is recommended. In situations where medication appears to be causing xerostomia, including instances of acidic medications such as inhalers or liquid or chewable tablets, the xerogenic medication should be replaced with one with less xerogenic side effects, if the medical conditions permit.
Diet should also be addressed in a preventive program. Patients should maintain a food diary for a week and analyze it to minimize acidic exposure in the oral cavity. Frequency and duration of introduction of acidity into the oral cavity must be reduced; drinking less acidic juices that are fortified with fluoride and minerals like calcium and phosphate should be encouraged. Consumption of neutralizing food, eg, cheese, should be suggested.
After consumption of food, rinsing the mouth with plain water, low-fluoride–containing rinse, milk, or baking soda (sodium bicarbonate) may help return pH to the normal range quickly. Also, brushing after acidic exposure in the oral cavity should be discouraged. Using less toothpaste and one with low relative dentin abrasivity on a soft toothbrush with round-ended filaments should be encouraged. Altering brushing technique and applying less force should also be suggested.
In the presence of bruxism, treatment becomes extremely difficult and restorations usually fail. Treatment of bruxism may involve behavior modification, such as stress management and relaxation therapy, eg, Yoga and other exercise, self-relaxation, and self-hypnosis. Mechanical devices such as mouthguards to protect the teeth from the forces of contact may also be effective.
Remineralizing the Tooth Structure
Every attempt should be made to remineralize the tooth surface and minimize the progression of tooth surface loss. Fluoride helps with the reuptake of available calcium and phosphate present in saliva. Fluorides are available in various concentrations and can be applied by the patient (prescription-strength toothpaste), eg, sodium fluoride (5,000 ppm), or professionally via varnishes (26,000 ppm). The reuptake of minerals can be enhanced by supplying or applying calcium and phosphate ions in various forms. Fluoride, sodium hexametaphosphates, ferrous sulphate, and stannous fluoride-containing mouthrinses work as anti-erosive agents.
Restoration of Teeth
Physiological tooth surface loss does not always require treatment. Once the tooth surface loss is determined to be pathological, however, the appropriate treatment is deemed necessary. Active treatment depends upon individual considerations and the needs of the patient, the degree of damage, the skill of the restorative dentist, time, and cost. Numerous reasons, based on accepted engineering principles, indicate that non-carious lesions should be restored.48 The etiological factors must be determined and controlled to initiate active dental restorations in order to confirm the success of the treatments rendered.
While restoration of worn teeth is easily done on non-occluding surfaces,50 the restoration of teeth with non-carious cervical lesions may not increase fracture resistance.51 These restorations require minimal tooth preparation and roughening of the lesions because these actions do not increase the retentiveness of the restorative materials.52 The primary cause of failure in non-carious cervical restorations is microleakage.53
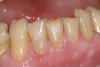
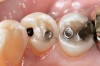
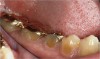
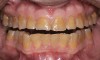
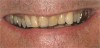
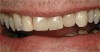
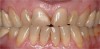
If the centric relation and centric occlusion in the presence of tooth surface loss is the same, or the dento-alveolar compensation has occurred, the teeth should be restored.54 The restorations can include Class V or palatal restorations, or reproduction of the cusps (Figure 9). Also, restorations can include covering the affected tooth surfaces with resin-based composites, using a sandwich technique, or placement of metallic, porcelain-fused-to-metal, or ceramic crowns (Figure 10, Figure 11, Figure 12, Figure 13, Figure 14, Figure 15 and Figure 16).
If the centric occlusion is not the same as centric relation, as in localized tooth surface loss that involves only lower anteriors, then monitoring should suffice. But if the localization is in the uppers, a Dahl type of appliance and restoration is preferred.54 A Dahl appliance is a metallic bite plate used to restore worn anterior teeth by creating an interocclusal space.55
If the freeway space has increased due to tooth surface loss, position of the centric relation should be established with a stabilization splint and the teeth restored to the existing facial height. If the freeway space is normal, ie, in dento-alveolar compensation, a stabilization splint should be used at increased vertical height. If the splints are tolerated then the restoration should be done at this increased vertical height; however, if they are not tolerated then a crown-lengthening procedure should be considered.54
Conclusion
Tooth surface loss and the resulting non-carious lesions are issues that can be difficult to diagnose correctly and treat successfully. The etiology of the tooth surface loss can be determined by reviewing the potential causes for each patient and by observing the pattern of wear, as the loss is exhibited in different patterns and on different surfaces of the teeth for varying types of tooth surface loss (ie, erosion, attrition, abrasion, and abfraction, or a combination). Determining the etiology and preventing further tooth surface loss, which requires using proper preventive methods, good management, and the appropriate restorations, is necessary for restorations to be successful.
References
1. Eccles JD. Tooth surface loss from abrasion, attrition and erosion. Dent Update. 1982;9(7):373-381.
2. Smith BG, Knight JK. An index for measuring the wear of teeth. Br Dent J. 1984;156(12):435-438.
3. Flint S, Scully C. Orofacial age changes and related disease. Dent Update. 1988;15(8):337-342.
4. Wilder-Smith CH, Wilder-Smith P, Kawakami-Wong H, et al. Quantification of dental erosions in patients with GERD using optical coherence tomography before and after double-blind, randomized treatment with esomeprazole or placebo. Am J Gastroenterol. 2009;104(11):2788-2795.
5. Joint Report of the American Dental Association Council on Access, Prevention and Interprofessional Relations and Council on Scientific affairs to the House of Delegates: Response to Resolution 73H-2000, October 2001.
6. Ganss C. Definition of erosion and links to tooth wear. In: Lussi A, ed. Dental Erosion: From Diagnosis to Therapy. Vol 20. Basel, Switzerland: S. Karger AG; 2006:9-16.
7. Barbour ME, Rees GD. The role of erosion, abrasion and attrition in tooth wear. J Clin Dent. 2006;17(4):88-93.
8. Grippo JO, Simring M, Schreiner S. Attrition, abrasion, corrosion and abfraction revisited. a new perspective on tooth surface lesions. J Am Dent Assoc. 2004;135(8):1109-1118.
9. Milosevic A. Tooth wear: an aetiological and diagnostic problem. Eur J Prosthodont Restor Dent. 1993;1(4):173-178.
10. Lussi A, Jaeggi T, Zero D. The role of diet in the aetiology of dental erosion. Caries Res. 2004;38(suppl 1):34-44.
11. Hara AT, Lussi A, Zero DT. Biological factors. In: Lussi A, ed. Dental Erosion: From Diagnosis to Therapy. Vol 20. Basel, Switzerland: S. Karger AG; 2006:88-99.
12. House RC, Grisius R, Bliziotes MM, Licht JH. Perimolysis: Unveiling the surreptitious vomiter. Oral Surg Oral Med Oral Pathol. 1981;51(2):152-155.
13. Harte DB, Manly RS. Effect of toothbrush variables on wear of dentin produced by four abrasives.J Dent Res. 1975;54(5):993-998.
14. Björn H, Lindhe J. On the mechanics of toothbrushing. Odontol Revy. 1966;17(1):9-16.
15. Litonjua LA, Andreana S, Bush PJ, et al. Wedged cervical lesions produced by toothbrushing. Am J Dent. 2004;17(4):237-240.
16. Philpotts CJ, Weador E, Joiner A. The measurement in vitro of enamel and dentine wear by toothpastes of different abrasivity. Int Dent J. 2005; 55(3 suppl 1):183-187.
17. Wiegand A, Schwerzmann M, Sener B, et al. Impact of toothpaste slurry abrasivity and toothbrush filament stiffness on abrasion of eroded enamel - an in vitro study. Acta Odontol Scand. 2008;66(4):231-235.
18. Volpe AR, Mooney R, Zumbrunnen C, et al. A long term clinical study evaluating the effect of two dentifrices on oral tissues. J Periodontol. 1975;46(2):113-118.
19. Pindborg JJ. Pathology of The Dental Hard Tissues. Philadelphia, PA: WB Saunders Co; 1970;274-320.
20. Grippo JO. Abfractions: a new classification of hard tissue lesions of teeth. J Esthet Dent. 1991;3(1):14-19.
21. McCoy G. The etiology of gingival erosion. J Oral Implantol. 1982;10(3):361-362.
22. Lee WC, Eakle WS. Possible role of tensile stress in the etiology of cervical erosive lesions of teeth. J Prosthet Dent. 1984;52(3):374-380.
23. Litonjua LA, Andreana S, Bush PJ, et al. Noncarious cervical lesions and abfractions: a re-evaluation. J Am Dent Assoc. 2003;134(7):845-850.
24. Christensen GJ. Treating bruxism and clenching. J Am Dent Assoc. 2000;131(2):233-235.
25. Xhonga, FA. Bruxism and its effect on the teeth. J Oral Rehabil. 1977;4(1):65-76.
26. Young WG, Khan F. Sites of dental erosion are saliva-dependent. J Oral Rehabil. 2002;29(1);35-43.
27. Howden GF. Erosion as the presenting symptom in hiatus hernia: A case report. Br Dent J. 1971;131(10):455-456.
28. Järvinen V, Meurman JH, Hyvärnen H, et al. Dental erosion and upper gastrointestinal disorders. Oral Surg Oral Med Oral Pathol. 1988;65(3):298-303.
29. Lussi A, Jaeggi T. Erosion—diagnosis and risk factors. Clin Oral Investig. 2008;12(suppl 1):S5-S13.
30. Meurman JH, Toskala J, Nuutinen P, Klemetti E. Oral and dental manifestations in gastroesophageal reflux disease. Oral Surg Oral Med Oral Pathol. 1994;78(5):583-589.
31. Holbrook WP. Furuholm J, Gudmundsson K., et al. Gastric reflux is a significant causative factor of tooth erosion. J Dent Res. 2009;88(5):422-426.
32. Lussi A, Kohler N, Zero D, et al. A comparison of the erosive potential of different beverages in primary and permanent teeth using an in vitro model. Eur J Oral Sci. 2000;108(2):110-114.
33. Lussi A. Jaeggi T. Erosion—diagnosis and risk factors. Clin Oral Investig. 2008;12(suppl 1):S5-S13.
34. West NX, Hughes JA, Addy M. The effect of pH on the erosion of dentine and enamel by dietary acids in vitro. J Oral Rehabil. 2001;28(9):860-864.
35. Bader JD, McClure F, Scurria MS, et al. Case-control study of non-carious cervical lesions. Community Dent Oral Epidemiol. 1996;24(4):286-291.
36. Padbury AD, Ash MM Jr. Abrasion caused by three methods of toothbrushing. J Periodontol. 1974;45(6):434-438.
37. Lewis R, Dwyer-Joyce RS, Pickles MJ. Interaction between toothbrushes and toothpaste abrasive particles in simulated tooth cleaning. Wear. 2004;257(3-4):368-376.
38. Drisko C, Henderson R, Yancy J. A review of current toothbrush bristle endo-rounding studies. Compend Contin Educ Dent. 1995;16(7):694-698.
39. Grabenstetter RJ, Broge RW, Jackson FL, Radike AW. The measurement of the abrasion of human teeth by dentifrice abrasives: a test utilizing radioactive teeth. J Dent Res. 1958;37(6):1060-1068.
40. Joiner A, Pickles MJ, Tanner C, et al. An in situ model to study the toothpaste abrasion of enamel. J Clin Periodontol 2004;31( 6):434-438.
41. Kiliaridis S, Johansson A, Haraldson T, et al. Craniofacial morphology, occlusal traits, and bite force in persons with advanced occlusal tooth wear. Am J Orthod Dentofacial Orthop. 1995;107(3):286-292.
42. Lambrechts P, Braem M, Vuylsteke-Wauters M, Vanherle G. Quantitative in vivo wear of human enamel. J Dent Res. 1989;68 (12):1752-1754.
43. Lussi A, Schaffner M. Progression of and risk factors for dental erosion and wedge-shaped defects over a 6-year period. Caries Res. 2000;34(2):182-187.
44. Shortall AC. Cavity cleansers in restorative dentistry. Preliminary results from an in vitro scanning electron microscope study. Br Dent J. 1981;150(9):243-247.
45. Pashley DH. Smear layer: biologic considerations. Oper Dent Suppl. 1984;3:13-29.
46. Addy M. Tooth brushing, tooth wear and dentine sensitivity—are they associated? Int Dent J. 2005;55(4 suppl 1):261-267.
47. Wickens JL. Tooth surface loss. 6. Prevention and maintenance. Br Dent J. 1999;186(8):371-376.
48. Grippo JO. Noncarious cervical lesions: the decision to ignore or restore. J Esthet Dent. 1992;4(suppl):55-64.
49. Chadwick RG, Mitchell HL, Cameron I, et al. Development of a novel system for assessing tooth and restoration wear. J Dent. 1997;25(1):41-47.50.
50. King PA. Tooth surface loss: Adhesive techniques. Br Dent J. 1999;186:321-326.
51. Osborne-Smith KL, Burke FJ, Farlane T, Wilson NH. Effect of restored and unrestored non-carious cervical lesions on the fracture resistance of previously restored maxillary premolar teeth. J Dent. 1998;26(5-6):427-433.
52. van Dijken JW. Durability of three simplified adhesive systems in Class V non-carious cervical dentin lesions. Am J Dent. 2004;17(1):27-32.
53. Van Meerbeek B, Perdigao J, Lambrechts P, Vanherle G. The clinical performance of adhesives. J Dent. 1998;26(1):1-20.
54. Davies SJ, Gray RJ, Qualtrough AJ. Management of tooth surface loss. Br Dent J. 2002;192(1):11-23.
55. Dahl BL, Krogstad O, Karlsen K. An alternative treatment in cases with advanced localized attrition. J Oral Rehabil. 1975;2(3):209-214.
About the Authors
Mabi L. Singh, DMD, MS
Associate Professor
Tufts University School of Dental Medicine
Boston, Massachusetts
Gerard Kugel, DMD, MS, PhD
Professor, Associate Dean for Dental Research
Tufts University School of Dental Medicine
Boston, Massachusetts
Britta Magnuson, DMD
Research Associate
Tufts University School of Dental Medicine
Boston, Massachusetts
Athena Papas, DMD, PhD
Professor
Tufts University School of Dental Medicine
Boston, Massachusetts